Skin Therapy / Clinicians

Electroporation is the term used when pulsed electrical fields, generated by the ePORE® and applied to tissue, render the cell membrane porous. The pulses vastly increase the porosity of the cell membrane, enabling greatly enhanced passive diffusion of molecules by the tumour. As a result, low doses of chemotherapy can be very effective due to the increase in porosity of the tumour cell membrane, the drug can passively diffuse into the cell and induce tumour cell death. The clinical utilisation of electroporation applied with a low dose chemotherapy (electrochemotherapy) to induce tumour cell death has broadened considerably over the last decade. As tumour tissue is several times more conductive than healthy tissue it absorbs more electrical energy, meaning that cancer cells can be destroyed whilst preserving the surrounding healthy tissue.
Examples of cutaneous and subcutaneous cancers that can be treated with CUTIS include:
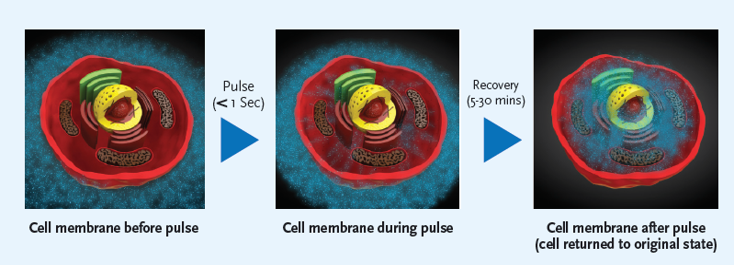
The application of pulsed electrical fields directly to tissue will create pores in the cell membrane in a process called ‘electroporation’. The pulses are created by the ePORE® generator and we refer to this treatment as ePORE® therapy. Effective treatment requires the electrical pulses to be above a threshold voltage with pulses of shorter duration requiring higher voltages to effect the membrane. Pulse lengths from nanoseconds to microseconds have been shown to impact on both internal organelles and the cell membrane respectively. The greater the number of pulses delivered the larger in number and size of pore created but this is generally limited to avoid heating of the tissue.
Pulsed electrical fields can be delivered alone to induce local tumour cell death in a process called Irreversible electroporation or when a lower voltage and pulse number are employed the membrane pore formation is reversible and this is typically combined with a drug or molecule such as calcium. These molecules are more easily absorbed by the cell via the pores created in the membrane after the pulsed electrical fields are generated. In the case where the pulsed electrical fields are combined with a chemotherapeutic agent the treatment is referred to as electrochemotherapy with the drug rendered far more effective due to its more direct absorption into the cancer cells. In the case of calcium, its level within the cell is carefully regulated and when an excess is absorbed as is the case after the application of pulsed electrical fields combined with the local injection of calcium into the tumour tissue it can trigger automatic cell death of the tumour.
The clinician will carefully consider the appropriate options for treatment considering the available clinical evidence, guidelines and the underlying health of the patient.
In the case of CUTIS and cutaneous or subcutaneous malignancies those typically considered suitable include:
ePORE® therapy and CUTIS can also be suitable for patients presenting with:
ePORE® therapy can be repeated for cutaneous and subcutaneous lesions as necessary, which is facilitated by its application under local anaesthesia.
Patients are typically treated as a day case, however, this must be assessed on a patient-by-patient basis and in some cases, it may be more appropriate to keep the patient under surveillance for the night after the procedure.
Some patients may experience a mild fever following the treatment, when it is combined with intravenous chemotherapy. Medication such as paracetamol can be prescribed to relieve this. On rare occasions, the wound site may become infected, and this is indicated by the appearance of increased redness or pain around the treated site, or discharge or fluid leakage. Your clinician will explain in detail the potential side effects and how they may be managed.
Serious side effects are extremely rare. In very few cases, patients may have an allergic reaction to the drug. The clinician responsible for your care will advise on the potential side effects and how they may be managed.
When utilised to manage late stage disease then the aim is purely quality of life improvement for the patient. For earlier stage disease and basal cell carcinoma the intent is curative and patients should discuss all the available options with their clinician.
ePORE® therapy is ideally suited where tissue preservation and local healing can be challenging post procedure.
For patients who present with larger lesions (>3cm), ePORE® therapy can greatly reduce the tumour and provide comfort and a greater quality of life.
If a patient has a pacemaker, then treatment should not be given in areas that are in close proximity to the pacemaker. Please make your clinician aware if you have a pacemaker and require treatment.
Very low concentrations of bleomycin are used and maintain their effectiveness due to the increased porosity of the tumour cells, thereby allowing maximum absorption of a low dose chemotherapeutic drug. If bleomycin is injected intravenously, the maximum dose is 15,000 IU/m2. If injected intratumorally, the dose is 1000 IU/ml.
There are very few side effects associated with the use of bleomycin in combination with ePORE® therapy, as the doses used are ten-fold lower than the standard doses used without electroporation.
In the case of calcium a concentration of 220 mmol has been utilised in clinical studies to date and is injected intralesionally just prior to electroporation. Please refer to our references section for further information.
